Coronavirus: Germany set to limit AstraZeneca jab to under-65s
Germany’s vaccine committee has said AstraZeneca’s Covid jab should only be given to people aged under 65.
The committee cited “insufficient data” for the effectiveness of the vaccine in those over 65.
Efficacy studies for the AstraZeneca vaccine have so far been carried out on a small sample of the elderly population, EU regulators have said.
The European Medicines Agency is to decide on Friday whether to approve the vaccine for use across the EU.
The UK has been using the AstraZeneca vaccine in its mass immunisation programme for weeks now, and public health officials say the vaccine is safe and provides “high levels of protection” against Covid-19.
The news comes with the EU in dispute with leading manufacturers over a shortage of vaccines on the continent.
UK-based AstraZeneca has said production issues at its Europe-based plants mean it will be unable to deliver the promised number of doses to the bloc.
But the EU says the firm must honour its commitments and deliver the jabs by diverting stock from the UK. Pfizer-BioNTech has also cut the number of dozes it is delivering to the 27-member bloc.
- What’s the problem with the EU’s vaccine programme?
- UK vaccine supplies ‘won’t be interrupted’
- EU vaccine delays prompt press frustration


All of the regulators and experts in different countries have been looking at the same data on the Oxford-AstraZeneca vaccine. That data comes from clinical trials, and those did recruit fewer elderly people overall.
That’s because they started off first with younger volunteers to get results as quickly as possible, given the urgency to find out if a vaccine would work well enough to help get us out of the pandemic.
The scientists who ran the trials have always been upfront about this. But they say there is other evidence to suggest the vaccine will work well in older adults. Studies show the over 65s have a strong immune responses to the vaccine. After receiving the shots their blood has plenty of the required antibodies that can fight coronavirus.
The UK has been using the AstraZeneca vaccine in its mass immunisation programme for weeks now and should soon have more proof from the real world setting about how much protection the shots give.

The independent vaccine commission advising the German government said on Thursday that there were “currently insufficient data available to assess the vaccine efficacy from 65 years of age” and recommended “the AstraZeneca vaccine… should only be offered to people aged 18-64 years at each stage”.
However, Dr Mary Ramsay, Head of Immunisations at Public Health England, said both the AstraZeneca and Pfizer-BioNTech vaccines are “safe and provide high levels of protection against Covid-19, particularly against severe disease.
“There were too few cases in older people in the AstraZeneca trials to observe precise levels of protection in this group, but data on immune responses were very reassuring.”
- The global plan to share Covid-19 vaccines fairly

Germany’s slow start turns political

Germany’s vaccination programme has got off to a slow start; just over two million doses have been administered since it began at Christmas.
It is relying, for now, on the US/German Pfizer-BioNTech vaccine as well as the Moderna jab. In the next few weeks, BioNTech is due to start producing more of its vaccine at a new site in the town of Marburg in western Germany.
It took workers not far away just days to construct a huge vaccination centre in Marburg. Inside there are widely spaced chairs, pot plants and rows of private cubicles where doses can be administered.
But, owing to recent supply and production issues, there are no vaccines. The centre – where staff could administer up to 1,200 doses a day – is still empty.
It’s not all bad news, however. Mobile teams in the area are now giving care home residents their second doses.
The issue has unsurprisingly become a political battleground. Germany’s government and in particular its health minister – who some see as a potential successor to Chancellor Angela Merkel – have come under fire for not securing more doses.

What’s happening with the rollout elsewhere?
The pace of the EU’s vaccination programme has been criticised in recent weeks, with a number of countries failing to hit their own targets. Officials have blamed a combination of logistical and supply issues.
In Spain, authorities in the capital Madrid have suspended all first doses of the vaccination for at least two weeks because of the ongoing shortages.
A number of other regions, including Catalonia in the north-east, are having similar problems with stock. Catalan officials believe their 30,000 remaining doses will run out this week.
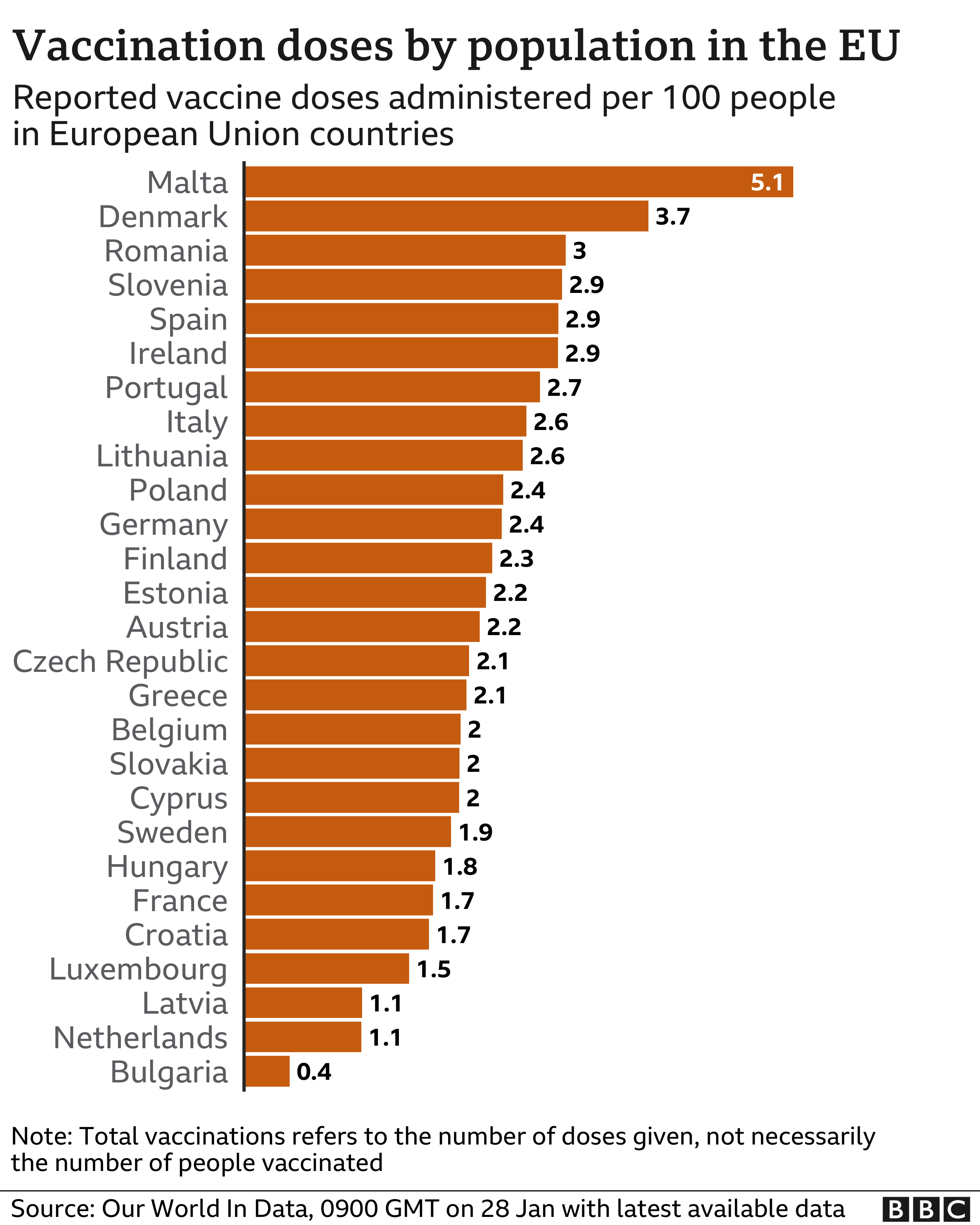

Many countries are facing similar shortages due to delays in shipments of both the Pfizer-BioNTech and Moderna vaccines, which are the only ones currently approved for use in the EU.
There was added confusion in the Czech Republic on Wednesday, after the health ministry called for a two-week halt to new vaccinations due to supply issues only for their statement to be denied by the prime minister.
“This [health ministry statement] was unnecessary,” Prime Minister Andrej Babis said. “The vaccination centres need to deal with this themselves.”
Elsewhere, Portugal is recording a high number of new infections and reports suggest hospitals in Lisbon are struggling to cope with the influx of patients.
A record 293 deaths were recorded in the country on Wednesday, and doctors from the German army have been deployed to Lisbon to help manage the spike.
Cases have also increased in parts of France and President Emmanuel Macron is reportedly mulling a stricter lockdown.
What’s behind the supply problems?
The EU signed a deal with AstraZeneca in August for 300 million doses, with an option for 100 million more, but the UK-Swedish company has reported production delays at plants in the Netherlands and Belgium.
AstraZeneca CEO Pascal Soriot said production was “basically two months behind where we wanted to be”.
The EU had hoped that as soon as approval was given – probably on Friday – delivery would start straight away, with some 80 million doses arriving in the 27 nations by March. But the production issues have dented this hope.
Reports said last week the EU would get 60% fewer vaccine doses – about 50 million jabs – than promised in the first quarter of the year.
The bloc is also facing delays with supplies of the Pfizer-BioNTech vaccine, and it has a much bigger deal with the US-German vaccine maker.
Both the EU and AstraZeneca have vowed to work together to resolve the problems, and crisis talks were held on Wednesday.
Published at Thu, 28 Jan 2021 14:53:45 +0000



Comments
Loading…